Basic Compliance Workflow for Cosmetics in Vietnam
Bycheermax
- On

1. Definition of “Cosmetic” under Vietnamese Law
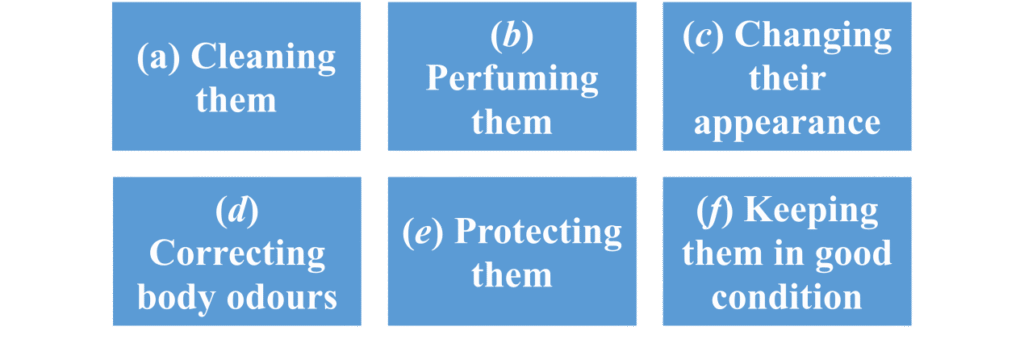
“A cosmetic product shall mean any substance or preparation intended to be placed in contact with the various external parts of the human body (epidermis, hair system, nails, lips and external genital organs) or with the teeth and the mucous membranes of the oral cavity with a view exclusively or mainly to cleaning them, perfuming them, changing their appearance and/or correcting body odours and/or protecting them or keeping them in good condition”
Products NOT regarded as cosmetics in Vietnam
-
- Mosquito repellents
-
- Fabric fragrances / laundry perfumes
-
- Fabric softeners
-
- Toilet-bowl cleaners
-
- Hydrogen-peroxide solutions
-
- 70 % or 90 % alcohol-based hand disinfectants
-
- Artificial-teeth cleaning products that do not contact the oral cavity
-
- etc.
2. Label requirements
-
- Product name and function
- Directions of use
- Full ingredient list (INCI, descending order)
- Country of origin
- Name and address of the Responsible Person in Vietnam
- Net content
- Batch number
- Date of manufacture or expiry (expiry date mandatory for shelf-life ≥ 30 months)
- Precautionary statements
- Inner package: at minimum product name + batch number
In addition to the basic requirements, also note:
-
- Directions of use, name/address of the company placing the product on the market and all precautionary statements must appear in Vietnamese.
- Language allowed: English and/or Vietnamese.
3. Claims
Vietnam refers to the basic requirements of ASEAN for claims:
-
- Permitted: conservative, realistic claims such as “cleansing”, “perfuming”, “improves appearance”, “protects”, “maintains good condition”.
-
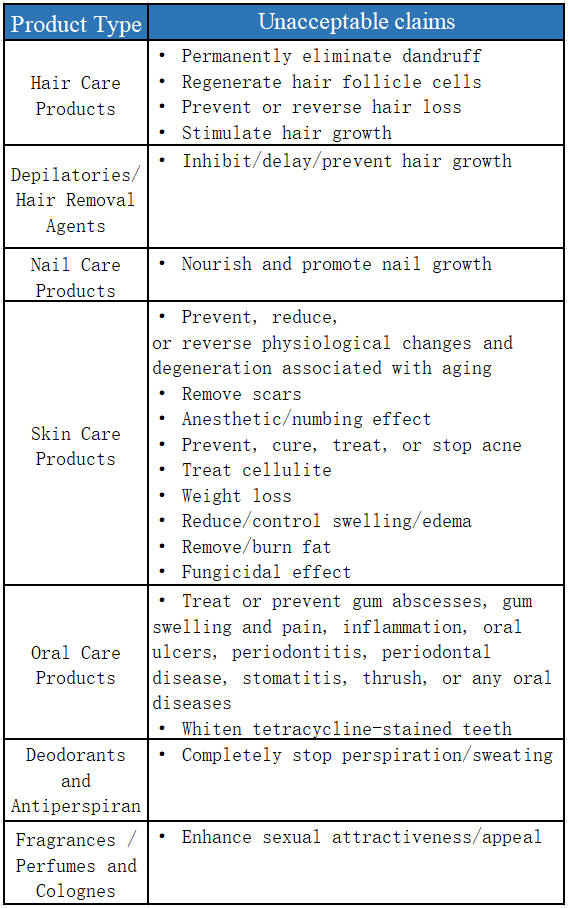
- Prohibited: medical or pharmacological terms and exaggerated/absolute statements, e.g. “permanently eliminates dandruff”, “removes scars completely”, “totally stops perspiration” or “enhances sexual attractiveness”.
4. Quality Requirements – Microbiological & Heavy-metal Limits
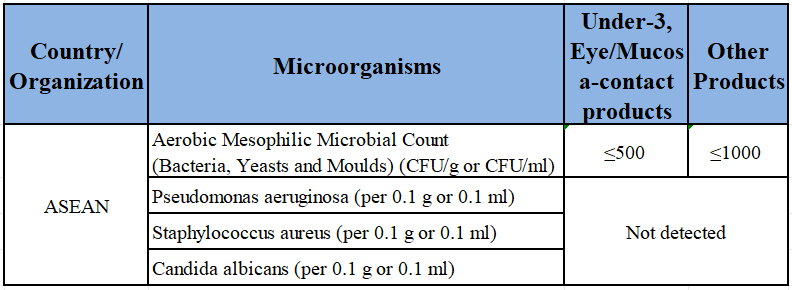
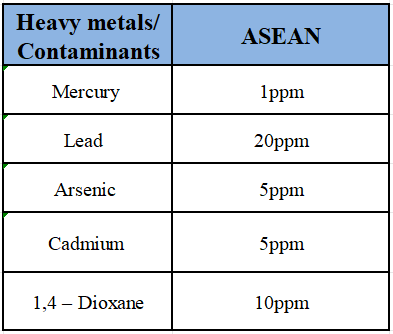
TIP: All limits apply to the FINISHED PRODUCT. Compliance at raw-material stage does not exempt the finished product.
-
- Testing method must be identified on COA or specification (e.g. USP, ISO, BP).
-
- Products containing water, plant extracts, talc or other high-risk ingredients: full-batch testing recommended.
-
- Low-risk formulations: quarterly testing acceptable if supported by stability data.
5. Notification Procedure
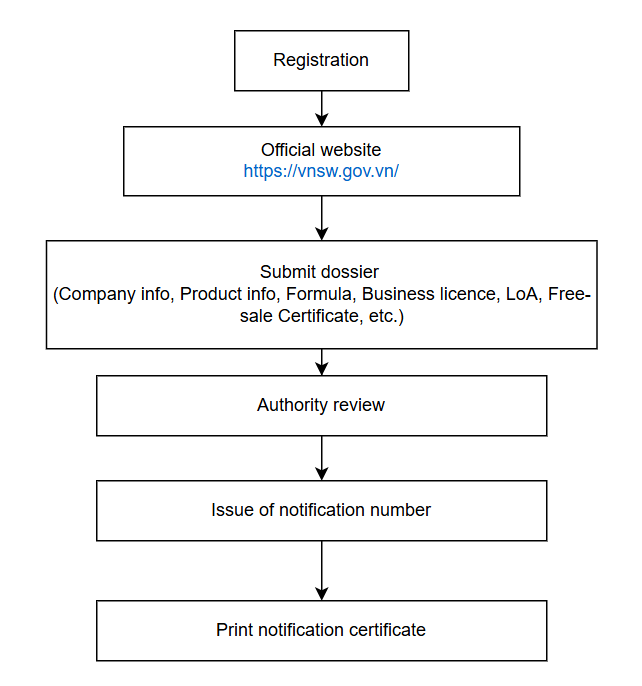
Registration Procedure
Only a legal entity established in Vietnam is eligible.
Allowed: 100 % Vietnamese-owned company, 100 % foreign-owned trading company, or a representative office.
Mainland Chinese or Hong Kong manufacturers cannot register directly; they must appoint a Vietnamese importer or set up a local subsidiary.
Portal for all applications: https://vnsw.gov.vn/
Information Request (Language: English or Vietnamese):
-
- Vietnam Company Certificate and Business License
- Manufacturer and Importer Information
- Product Formulation
- Product Information
- Authorization Letter (to be notarized and certified by Embassy)
- Certificate of Free Sale (to be notarized and certified by Embassy)
When uploading product information you need to fill out a TEMPLATE FOR NOTIFICATION OF COSMETIC PRODUCT which includes:
-
- Brand name
- Product name
- Model / shade list
- Product category
- Intended purpose
- Presentation form
- Manufacturer details
- Packaging-site details
- Details of the organisation/individual placing the product on the market
- Legal representative of the company
- Importer information
- Complete ingredient list with percentages for any restricted substances
Fee & Timeline
Fee: VND 500 000 (≈ CNY 140)
Administrative review by DAV: 5 working days
If supplementary information is requested, the applicant has 72 h (weekends/public holidays excluded) to upload the missing documents via the link provided in the DAV e-mail.
Failure to meet the deadline = automatic rejection, fee non-refundable.
After approval
A 5-year notification number is issued, format:
00012/CBMP-TB/2025
-
- 00012 = annual serial number (≤ 7 digits)
-
- CBMP = acronym for cosmetic notification
-
- TB = imported product (ĐK = locally manufactured)
-
- 2025 = year of issuance
Renewal: log in to the same portal 90 days before expiry; no re-evaluation of the formula is required if product, label and responsible company remain unchanged. New number usually issued within 7–10 working days.
6. Advertising
All cosmetic advertising in Vietnam (any media) must comply with Vietnamese law and obtain prior approval.
-
- Advertising application dossier
-
- Cosmetic notification form
-
- Copy of the cosmetic product notification certificate (company stamp required)
-
- Copy of the enterprise registration certificate (stamped)
-
- Authorisation letter from the product owner to the advertising applicant (notarised & legalised for imports)
-
- Scripts, images, music, video scenes, etc.
-
- Supporting evidence if advertised attributes differ from those declared in the notification
-
- Free-Sale Certificate (notarised & legalised for imports)
Prepare every detail in advance—the Vietnamese customs green light is waiting for you!

